The Quick Answer
The Quick Answer
The short answer is yes: titanium is a conductive metal.
However, if you are planning to use titanium to replace copper wiring in a project, you should stop. While titanium does conduct electricity, it is not a good conductor. In the world of metals, it is effectively a resistor.
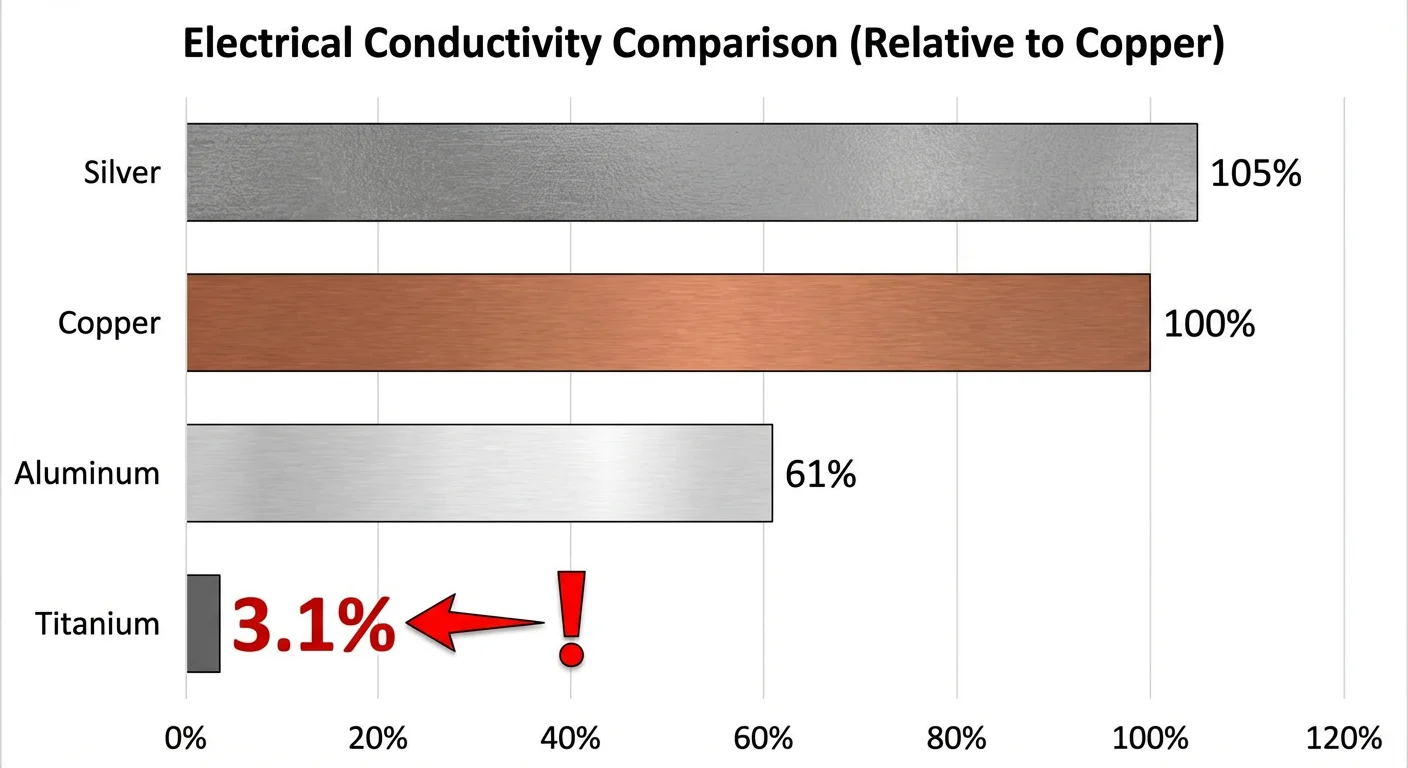
To give you a clear perspective on just how “poor” its conductivity is, we use the IACS (International Annealed Copper Standard) scale. If we treat Copper as the standard at 100%, Titanium scores only about 3.1%.
This means titanium has very high electrical resistance. When current passes through it, the metal resists the flow of electrons, causing it to heat up rapidly rather than transmitting the power efficiently.
So, is it useless for electronics? Absolutely not. This unique combination—being conductive enough to carry a charge but resistive enough to generate heat—makes titanium perfect for specific niche applications like vaping heating elements, anodized jewelry, and chemical processing equipment, even if it’s terrible for power cords.
How Conductive is Titanium? (The Real Numbers)
To understand exactly where titanium stands, we look at the IACS (International Annealed Copper Standard). This is the global benchmark for electrical conductivity, where pure copper is set at 100%.
If you look at the comparison below, the gap between titanium and standard conductive metals is massive:
| Metal | Conductivity (% IACS) | Performance Rating |
|---|---|---|
| Silver | 105% | Excellent |
| Copper | 100% | The Standard |
| Aluminum | 61% | Good |
| Titanium (Grade 1) | ~3.1% | Poor |
| Stainless Steel (304) | ~2.5% | Very Poor |
As you can see, titanium conducts electricity about 30 times worse than copper.
For engineers, this means titanium has high electrical resistivity (approximately 560 nΩ·m at 20°C). If you tried to use a titanium wire to power a device, you would experience a massive voltage drop. The energy that should be powering your device would instead be lost as heat along the wire.
Why is Titanium a Poor Conductor?
You might wonder: If it’s a metal, why doesn’t it let electricity flow freely?
The answer lies in its atomic structure.
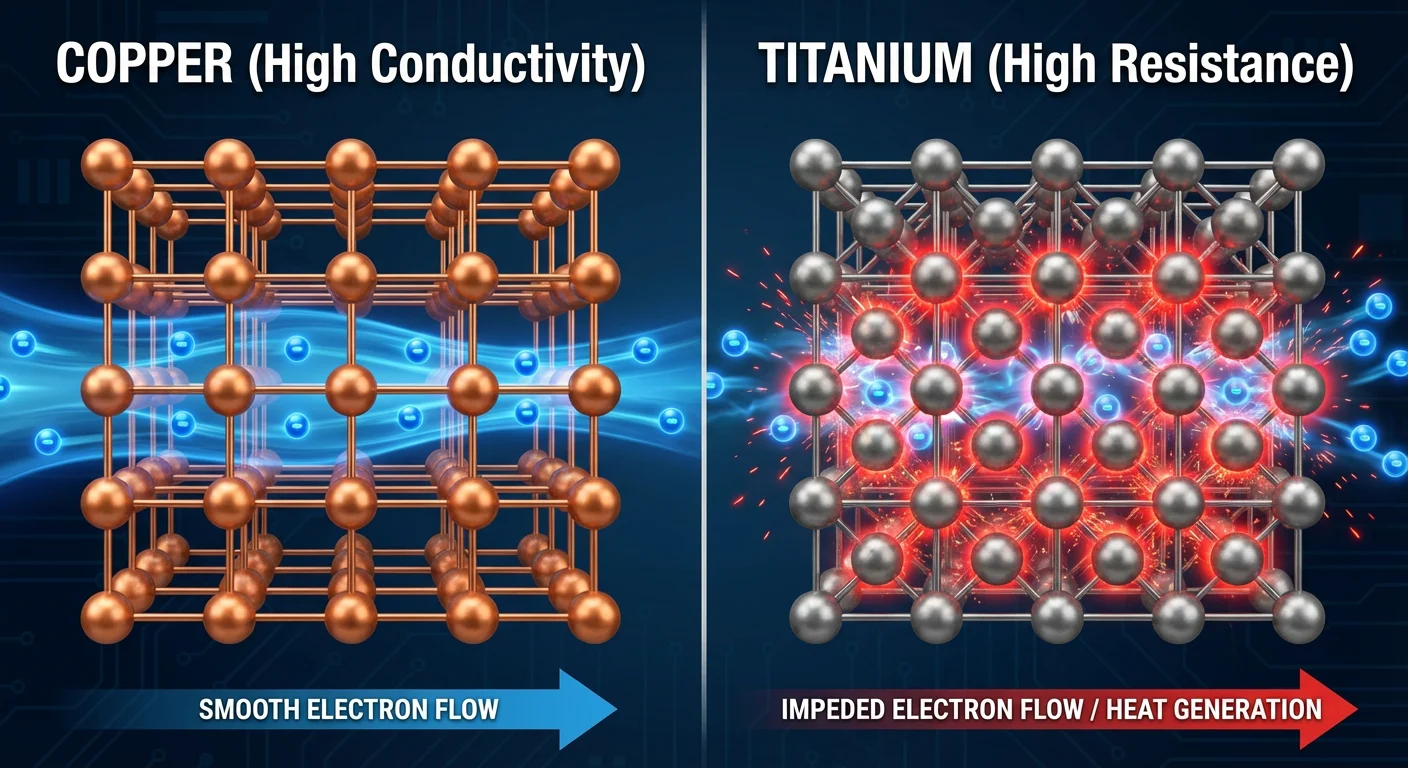
In good conductors like copper and silver, the outer electrons (valence electrons) are loosely held and can move freely through the metal’s crystal lattice. This “sea of electrons” allows current to pass with very little resistance.
Titanium, however, is a transition metal with a different electron configuration. Its outer electrons are more tightly bound, and its crystal structure creates more “friction” for moving electrons.
In physics terms, this friction is resistance. When you force electricity through titanium, the electrons collide frequently with the atoms, converting the electrical energy into thermal energy (heat).
This is exactly why titanium is terrible for wiring, but excellent for heating elements.
3 Ways We Use Titanium’s Electrical Properties
In engineering, there is no such thing as a “bad” property, only the wrong application. Titanium’s high resistance and unique electrical behavior make it the star material in these three specific scenarios:
1. Anodizing: Painting with Electricity
If titanium were not conductive, we wouldn’t have those vibrant, rainbow-colored titanium rings and knife scales.
The coloring process, known as Type III Anodizing, relies entirely on electricity. By submerging titanium in an electrolyte bath and running a current through it, an oxide layer forms on the surface.
Here is the cool part: Titanium’s resistance helps control this oxide growth perfectly. By changing the voltage, we change the thickness of the oxide layer, which refracts light differently to create specific colors:
- 30 Volts: Blue
- 55 Volts: Gold
- 75 Volts: Pink / Purple
- 100 Volts: Green
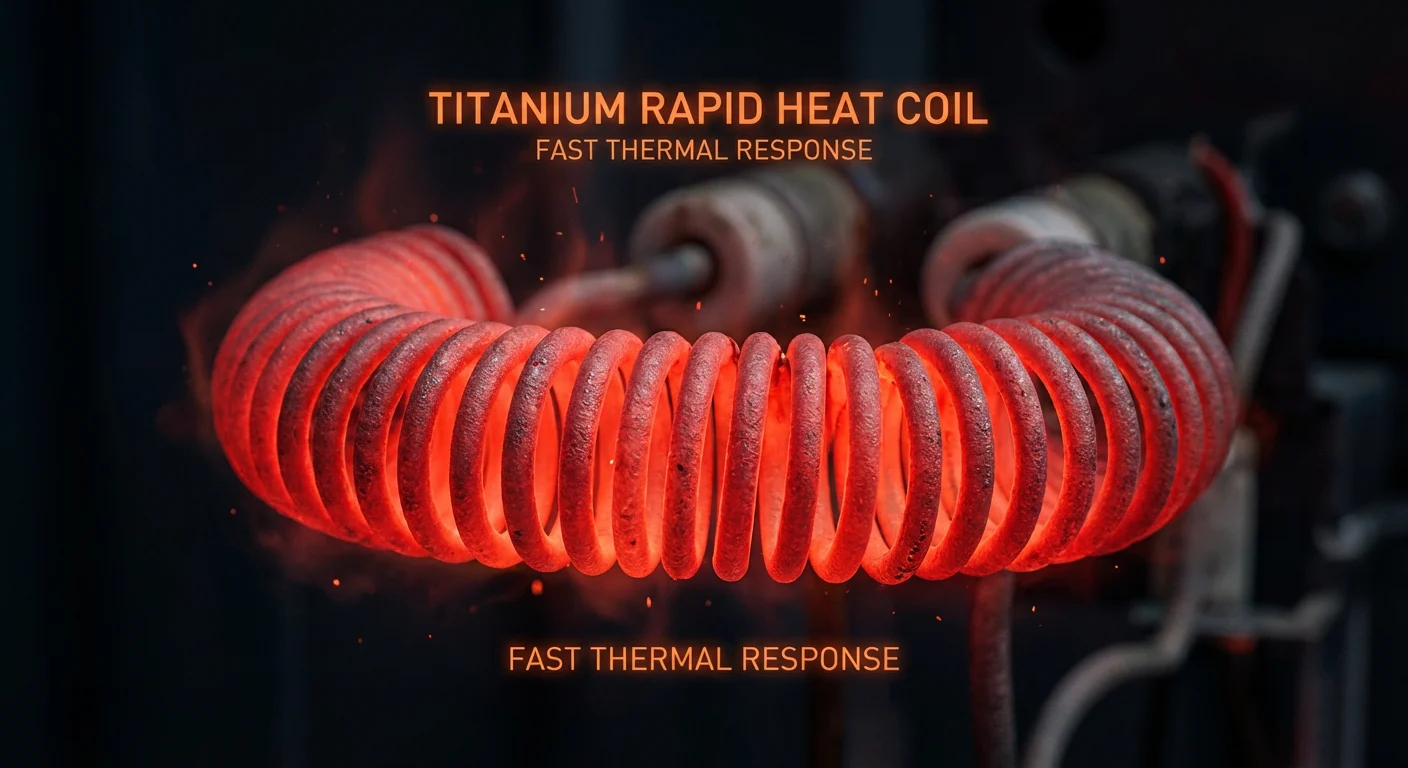
2. Vaping & Heating Elements (The “Joule Heating” Effect)
In the vaping community, Titanium (specifically Grade 1) is a popular choice for heating coils. Why? Because of its High Resistance and TCR (Temperature Coefficient of Resistance).
Since titanium resists electricity, it generates heat incredibly fast (Joule Heating). More importantly, its resistance changes predictably as it gets hotter.
- Safety Note: This is why vapers use titanium wire exclusively in “Temperature Control” (TC) mode. The chip reads the resistance change to calculate the exact temperature, preventing the wire from overheating and producing harmful oxides.
3. Impressed Current Cathodic Protection (ICCP)
This is a heavy industrial use. Because titanium is conductive but chemically inert (it doesn’t corrode easily), it is often used as the anode in systems designed to protect other metals from rusting.
For example, in reinforced concrete or seawater pipelines, titanium ribbons conduct electricity to “push” corrosion away from the steel structures, sacrificing nothing in the process thanks to its oxide film.
Warning: The Hidden Risk of Galvanic Corrosion
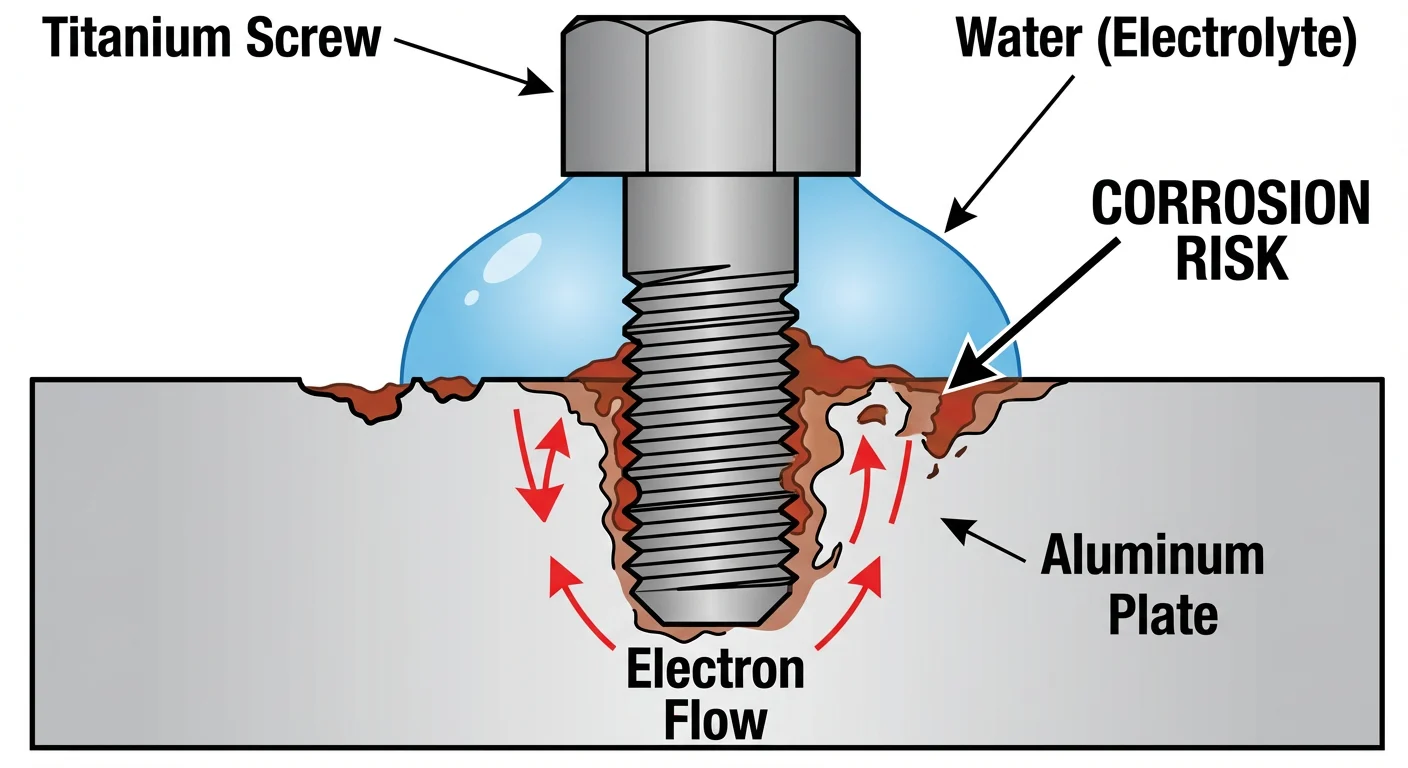
While titanium itself is immune to rust, its conductivity can actually destroy other metals around it. This phenomenon is called Galvanic Corrosion.
Because titanium is a conductive metal, when it touches a less noble metal (like Aluminum or Carbon Steel) in the presence of an electrolyte (like saltwater or sweat), it creates a miniature battery.
In this electrical circuit:
- The Titanium acts as the Cathode (Noble/Protected).
- The Aluminum acts as the Anode (Active/Sacrificial).
The result? The titanium stays shiny and new, but it accelerates the corrosion of the aluminum part, causing it to rot away much faster than normal.
The Fix: If you are designing a project that mixes titanium with other metals (like using titanium bolts on an aluminum bicycle frame), always use insulating paste, washers, or coatings to break the electrical connection.
Common Questions About Titanium
Here are the quick answers to the most confusing questions about titanium’s properties.
A: No. Titanium is paramagnetic. This means it is very weakly attracted to magnetic fields, but for all practical purposes (like sticking a magnet to it), it is non-magnetic. Being conductive does not mean being magnetic.
Q: Does titanium conduct heat well?
A: No. Just like its electrical conductivity, its thermal conductivity is also very poor (about 15x lower than aluminum). This is why titanium camping mugs don’t burn your lips instantly, and why titanium handles feel “warmer” to the touch in winter than steel.
Q: Is titanium a semiconductor?
A: No. Titanium is a metal and a conductor. However, its oxide (Titanium Dioxide, TiO2) is a semiconductor, which is widely used in solar cells and photocatalysts. But the metal itself is strictly a conductor.
Conclusion
Is titanium a conductive metal? Yes. But you should think of it more as a resistor than a conductor.
Its low conductivity (3.1% IACS) makes it a terrible choice for power transmission, but an incredible material for specialized applications where corrosion resistance, heating efficiency, and weight matter more than the flow of electrons.
References
To ensure accuracy and reliability, the data presented in this guide is referenced from the following authoritative industry standards and databases:
- MatWeb: Material Property Data for Titanium Alloys (Ti-6Al-4V & CP Grade 1).
- ASM International: Properties and Selection: Nonferrous Alloys and Special-Purpose Materials.
- ASTM B265: Standard Specification for Titanium and Titanium Alloy Strip, Sheet, and Plate.

 The Quick Answer
The Quick Answer